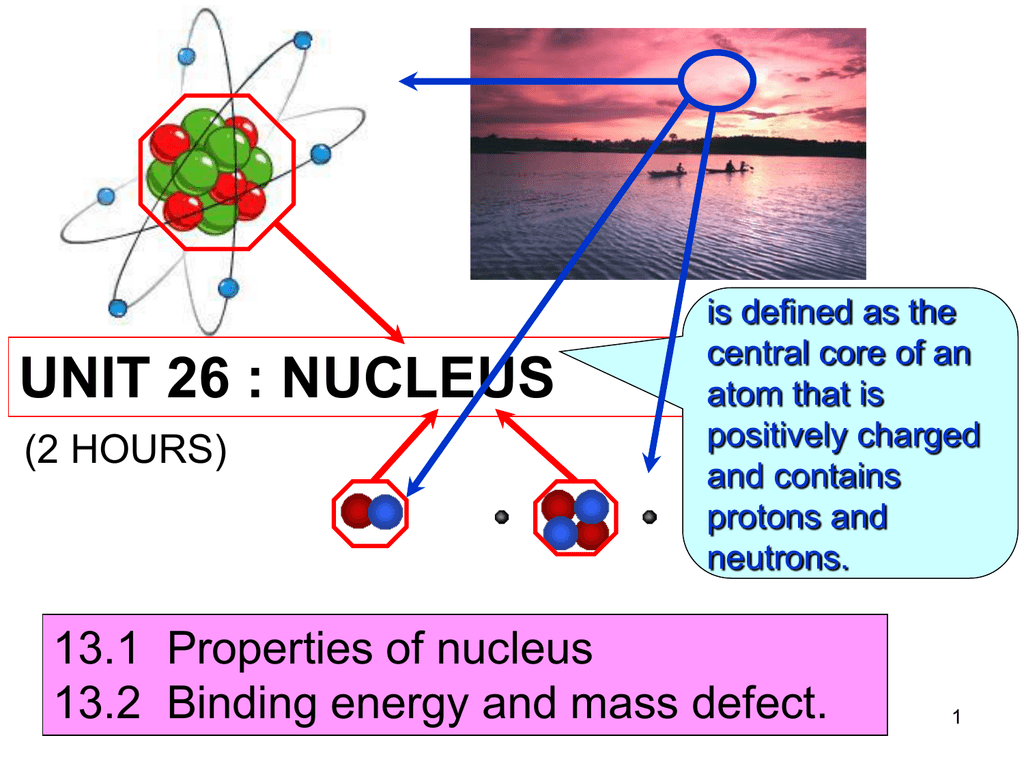
If M(A, Z), Mp and Mn denote the masses of the nucleus, proton and neutron respectively in units of u(1u = 931.5 MeV/c^2) and BE represents its binding energy in MeV, then.

m_(p) and m_(n)` are masses of proton and neutron respectively. An element of mass M has Z protons - YouTube

SOLVED: Semi-empirical mass formula: 2/3 22 (N-2)? B= #lap A 3/4 ahA-a,A a a, A1/3 ah =15.5 MeV a,=16.8 MeV 23 MeV ap = 34 MeV +1 1 = Zeven-N even nuclei

SOLVED: The following masses are given: 1 n 1.008665 0 1 H 1.007825 u 1 226 Ra 226.025403 u 88 The binding energy of 226 Ra, in MeV, is closest to which value? 88 2500 2100 1900 2300 1700
![SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses](https://cdn.numerade.com/ask_previews/6e66e007-83c2-43b6-af02-7fb376485580_large.jpg)
SOLVED: Question 2: [Marks: 4+6 ] (a) Explain as [0 why or why not the formula for total mass of a nucleus mw given below in term of proton and neutron masses

If M(A, Z), Mp and Mn denote the masses of the nucleus, proton and neutron respectively in units of u(1u = 931.5 MeV/c^2) and BE represents its binding energy in MeV, then.

Nuclear Phenomenology 3C24 Nuclear and Particle Physics Tricia Vahle & Simon Dean (based on Lecture Notes from Ruben Saakyan) UCL. - ppt download

and decays, Radiation Therapies and Diagnostic, Fusion and Fission This Lecture: Radioactivity, Nuclear decay Radiation damage, radiation therapies. - ppt download

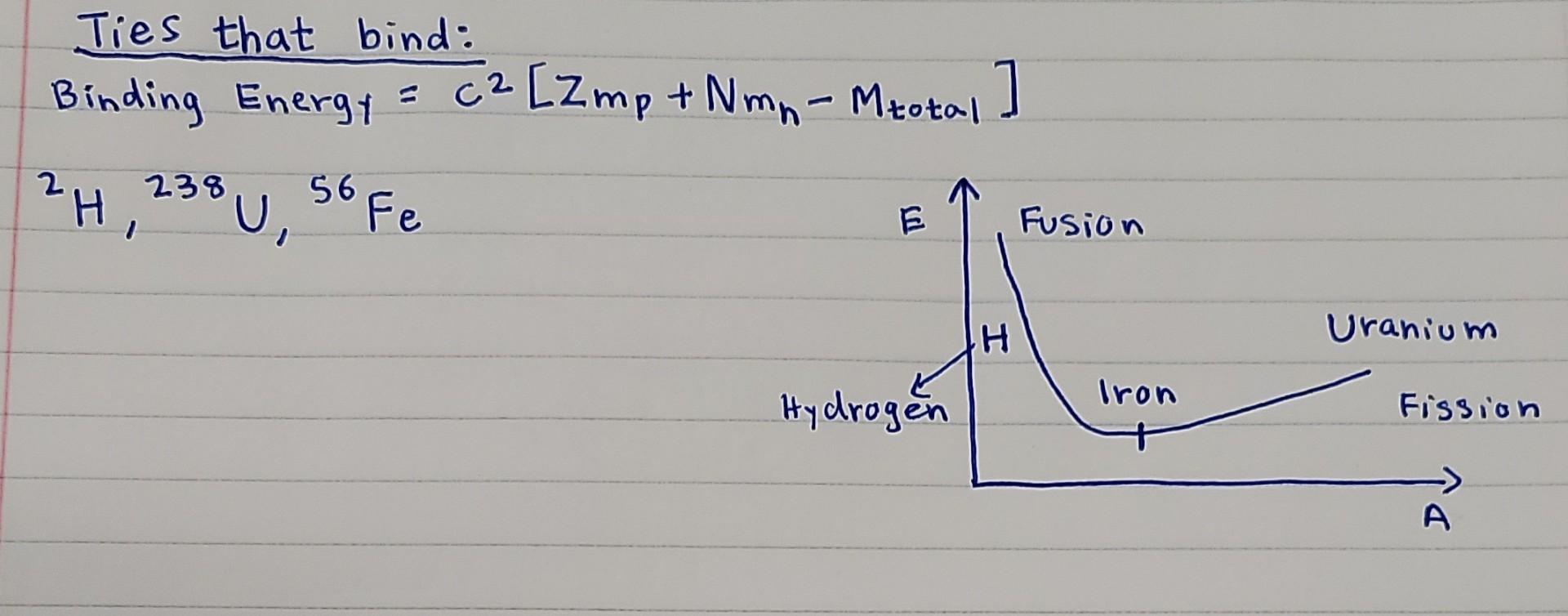
M, M n and M p denotes the masses of a nucleus of ZX^A , a neutron, and a proton respectively. If the nucleus is separated into its individual protons and neutrons

M, M n and M p denotes the masses of a nucleus of ZX^A , a neutron, and a proton respectively. If the nucleus is separated into its individual protons and neutrons


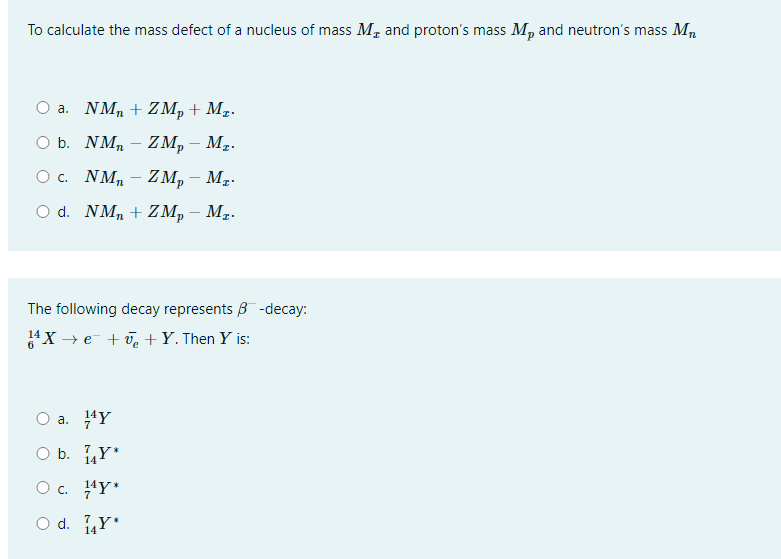



![Solved [Total marks:20] Toto QUESTION 3 Open with Google | Chegg.com Solved [Total marks:20] Toto QUESTION 3 Open with Google | Chegg.com](https://media.cheggcdn.com/media/1c6/1c6f7aed-79cb-4510-93f3-807974503ee4/phpNFa8ys.png)